Review | Open Access
From slides to signatures: pathological determinants of immunotherapy response in lung cancer
Omnia Mohamed Attia
Department of Medicine, Faculty of Medicine, Aswan University, Aswan 81528, Egypt.
Correspondence: Omnia Mohamed Attia (Department of Medicine, Faculty of Medicine, Aswan University, Aswan 81528, Egypt; Email: omnia992007a@gmail.com).
Asia-Pacific Journal of Oncology 2025, 6: 82-94. https://doi.org/10.32948/ajo.2025.11.15
Received: 15 Oct 2025 | Accepted: 20 Nov 2025 | Published online: 22 Dec 2025
Key words lung cancer, immune checkpoint inhibitors, PD-L1, cytotoxic T cells, tertiary lymphoid structures, circulating biomarkers, systemic inflammation indices
Predictive biomarkers represent measurable biological features to help estimate the probability of response to immune checkpoint blockade. Most importantly, they can help to identify those patients more likely to benefit from immune checkpoint inhibitors, which aids in appropriate individual treatment selection and can lead to improved safety and overall clinical benefit [7]. At the same time, clarifying the biology behind these markers can inform rational combination strategies, particularly for addressing primary resistance or relapses after an initial response. Therefore, not only does biomarker research strengthen the practical use of immune checkpoint inhibitors in the clinic, but it is a foundation for the next step in the progress of cancer immunotherapy, not without clinical and scientific value [8]. In addition, stratifying patients based on biomarkers may lower unnecessary treatment expenditures and promote more efficient use of healthcare resources [9]. The first major category of biomarkers of immunotherapy response is molecular biomarkers, which include PD-L1 expression, genetic mutations and genomic instability features, epigenetic biomarkers and transcriptomic signatures [10]. Next, cellular biomarkers of immunotherapy response are mainly attributed to different kinds of tumor-infiltrating immune cells such as cytotoxic T cells, B-cells and immune inhibitory cells as well as the development of tertiary lymphoid structures [11]. Lastly, systemic biomarkers of immunotherapy response involve circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), circulating immune cells and their associated systemic inflammatory indices [12]. In this review, we discuss the recent progress made in establishing these biomarkers of immunotherapy response in lung cancer and highlight their potential to distinguish immunotherapy responders from nonresponders, thereby aiding in efficiently limiting lung cancer tumor burden.
PD-L1 inhibits T-cell-mediated antitumor immunity through its interaction with the PD-1 receptor. Immune checkpoint inhibitors work by disrupting this inhibitory signaling axis. As a result, higher PD-L1 expression is often linked to greater sensitivity of tumors to immune checkpoint inhibitors [13]. Because PD-L1 testing is comparatively well standardized and can be implemented routinely in clinical workflows, PD-L1 expression has become one of the most practical and commonly used biomarkers for predicting immune checkpoint inhibitor benefit, regardless of smoking status (Figure 1) [14]. In practice, PD-L1 can be measured using several scoring frameworks, including the tumor proportion score (TPS), tumor cell score, immune cell score, and combined positive score [15]. Available data suggest that the concomitant consideration of TPS along with combined positive score may refine performance from the prediction point of view more than either of the metrics [16]. Notably, early clinical findings have been shown to correlate PD-L1 expression above 5% with better responses to treatment with the PD-1 inhibitor nivolumab as early as in 2012 [17]. In one study in patients with NSCLC treated with PD-1 blocking antibody (pembrolizumab), patients with a PD-L1 TPS >50% showed an objective response rate of 45.2% [18]. Moreover, compared to chemotherapy, treatment with pembrolizumab in TPS patients with PD-L1>50% led to significantly prolonged overall and progression-free survival and lowered the incidence of adverse events [19, 20]. These findings resulted in approval of nivolumab by the FDA in 2016 as a first-line therapy or therapy following the initial treatment for NSCLC with the expression of PD-L1 factored into treatment decision-making explicitly [21]. In the CheckMate 227 trial, a PD-L1 expression threshold of 1% was used, while patients with PD-L1 expression >1% and PD-L1 expression <1% had a 5-year survival rate of 24% versus 14% and 19% versus 7% in combination immunotherapy versus chemotherapy, respectively [22]. In a similar setting, patients with NSCLC who underwent concurrent chemoradiotherapy followed by durvalumab demonstrated an overall survival benefit mainly among PD-L1 expression ≥1% [23]. However, despite the clear value in the clinic, PD-L1 expression is marked by temporal and spatial heterogeneity, and therefore discordance may exist between primary lesions and metastatic sites. In addition, the most appropriate threshold for defining PD-L1 positivity remains controversial [24]. Therefore, PD-L1 status alone may not provide sufficient predictive power, and its combination with other biomarkers represents a better approach for anticipating immune checkpoint inhibitor responses.
Genetic biomarkers
Genomic instability is a hallmark of malignant transformation and arises from alterations within the tumor cell genome. Mismatch repair deficiency and high microsatellite instability are uncommon in NSCLC; however, NSCLC exhibits the highest overall tumor mutational burden [25]. The broad distribution of tumor mutational burden in NSCLC is partly explained by distinct mutational landscapes in smokers versus never-smokers, which generate substantially different somatic mutation profiles [26]. Tumor mutational burden serves as a predictive biomarker for immune checkpoint inhibitor benefits in NSCLC, and this association has been reported even when accounting for PD-L1 expression status (Figure 1) [27]. In addition to overall mutation load, chromosomal instability may also shape immunotherapy outcomes. In immunotherapy-treated NSCLC cohorts, tumors with high copy number variation were reported to show higher expression of PD-L1, CD39, and CD19, together with increased infiltration of CD8⁺ and CD3⁺ T cells. This profile is consistent with a more immune-active tumor state and suggests that copy number variation could serve as an additional genomic indicator for immunotherapy responsiveness in NSCLC [28]. Oncogenic driver alterations further influence immune checkpoint inhibitor efficacy in NSCLC by affecting PD-L1 expression, tumor mutational burden, and the tumor microenvironment. For instance, epidermal growth factor receptor (EGFR)-mutant NSCLC is often characterized by lower tumor mutational burden and reduced PD-L1 expression, while KRAS-mutant tumors more frequently show higher levels of both. The most common EGFR alterations, exon 19 deletions and the exon 21 L858R substitution, together represent more than 90% of EGFR mutations and can substantially reprogram the tumor microenvironment. Mechanistically, EGFR signaling can activate the PI3K-AKT-mTOR pathway and increase expression of chemokines, which shifts the dynamics towards immunosuppressive environment [29, 30]. In line with this, NSCLC subsets harboring ALK, EGFR, HER2, or RET alterations generally derive limited benefit from PD-1/PD-L1 blockade when used alone. On the other hand, tumors with concurrent TP53 and KRAS mutations show better response to immune checkpoint inhibitors, while those with KRAS plus STK11 and/or KEAP1 co-mutations are frequently resistant to immunotherapy (Figure 1) [29].
Epigenetic biomarkers
Epigenetic changes contribute to tumor initiation and progression and affect responsiveness to immune checkpoint inhibitors [31]. Through mechanisms like DNA methylation and histone modification, epigenetic programs can be used to control immune checkpoints, which can influence the outcome of immunotherapy [32]. For example, genome-wide epigenetic modification patterns defined by methylation in promoter regions can predict immunotherapy response in NSCLC [33]. In stage IV patients with NSCLC treated with anti-PD-1 agents, a classifier of DNA methylation results, which was called EPIMMUNE, was linked with both progression-free and overall survival [34]. Histone deacetylases (HDACs) represent another epigenetic axis relevant to antitumor immunity. In the case of NSCLC, the anti-proliferative HDAC8 has been reported to be able to restore CD8+ T-cell effector function and improve responses to anti-PD-1 therapy (Figure 1) [35]. In parallel, HDAC6 expression has been proposed as a potential prognostic marker in patients undergoing immune checkpoint inhibitor treatment, and preclinical data provide evidence that the combination of inhibition of histone deacetylase enzyme family with inhibition of PD-1 checkpoint protein can inhibit tumor growth, and also favors a tumor microenvironment that is better conducive to the activity of cytotoxic T lymphocytes [36]. Beyond tissue-based profiling, integrating the epigenetic markers into the liquid biopsy platforms is a new frontier. DNA methylation features observed in ctDNA can be potentially useful for preliminary detection, treatment monitoring and response prediction [37]. However, although a correlation between ctDNA methylation and outcomes for targeted therapy has been established, its predictive value for the efficacy of immune checkpoint inhibitor is less clear and requires further validation [37]. Overall, epigenetic control is closely linked to antitumor immunity, and further research could provide clinically useful biomarkers in lung cancer.
Transcriptomic biomarkers
Transcriptomic profiling-based biomarkers provide a powerful framework to predict immune checkpoint inhibitor responses. By capturing transcriptional programs related to T-cell activation, antigen presentation, and interferon-γ (IFNγ) signaling, transcriptomic signatures can provide a functional readout of tumor immune status [38]. IFNγ is a central antitumor immunity regulator and is tightly associated with PD-L1 expression. In several cancer types, expression of mRNA related to IFNγ has been shown to be predictive of responses to PD-1 blockade (Figure 1) [39]. In contrast, the resistance to immune checkpoint inhibitors has been linked to genetic alterations that involve components of the IFNγ signaling cascade [6]. In patients treated with Atezolizumab, higher expression of T cell effector genes and IFNγ related programs had the association with an extended overall survival [40]. IFNγ expression itself has also been associated with benefit with the median progression-free survival being significantly longer in patients in the nivolumab group with higher levels of IFNγ [41]. Composite transcriptomic models may further strengthen predictive performance. The GDPLichi score, constructed from seven DNA damage repair-related genes, stratifies lung adenocarcinoma with the high-risk group showing increased tumor mutational burden, higher neoantigen load, and elevated expression of PD-L1 and CTLA4, suggesting greater immunotherapy sensitivity [42]. Additional transcriptomic markers, including CSF1R and HCST, have shown positive associations with PD-L1 expression and CD8⁺ T-cell infiltration, indicating potential prognostic and predictive value in NSCLC patients treated with anti-PD-L1 therapy [43]. Moreover, T-cell receptor co-expression gene signatures have been validated as predictors of immune checkpoint inhibitor benefit, with higher expression levels associated with improved outcomes (Figure 1) [44].
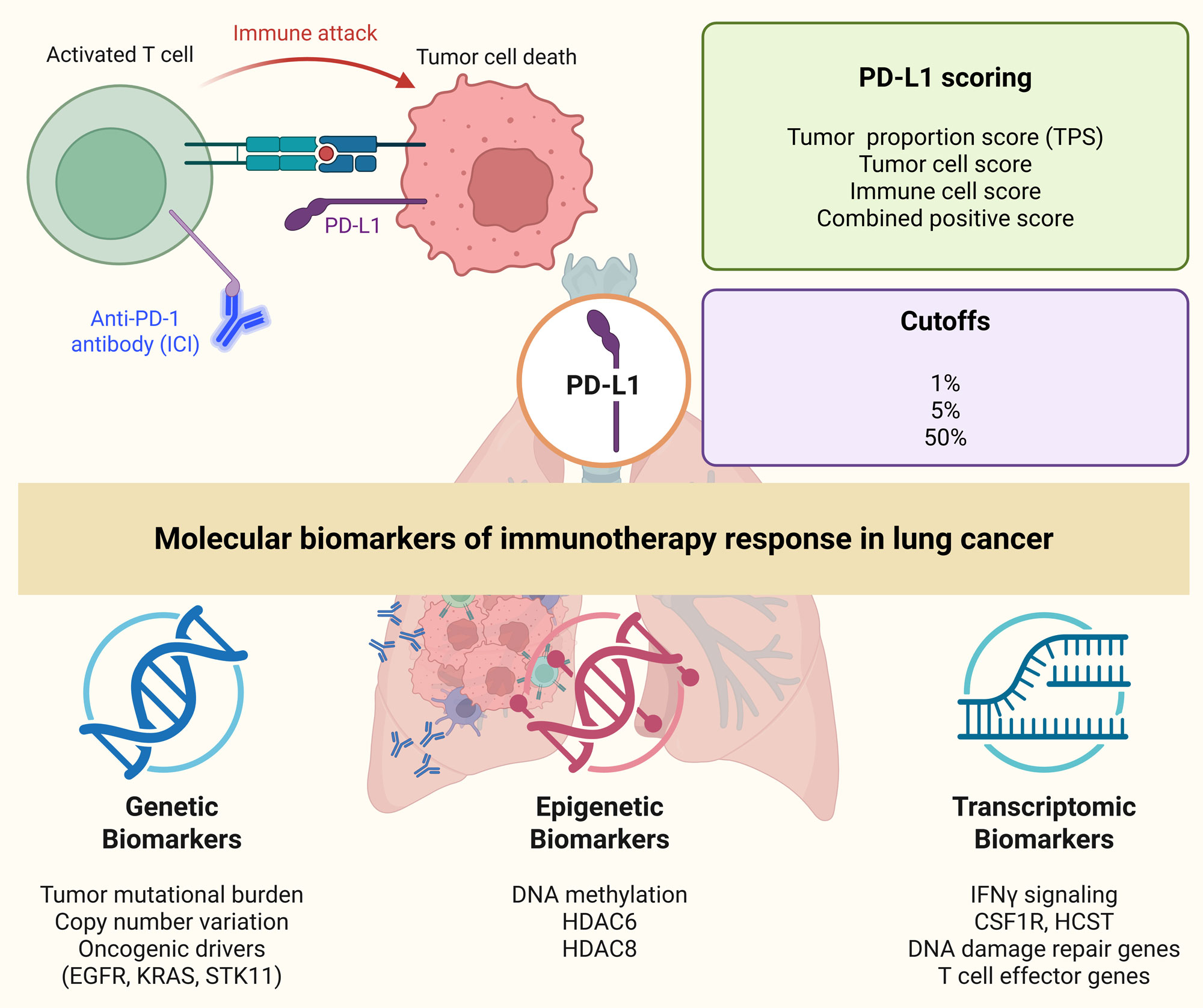 Figure 1. Molecular biomarkers of immunotherapy response in lung cancer. PD-1 blockade with immune checkpoint inhibitors (ICIs) restores T cell activity against tumor cells by disrupting the interaction between PD-1 and PD-L1 on T and tumor cells, respectively. Key molecular biomarkers include PD-L1 expression, assessed through various scoring methods (tumor proportion score, tumor cell score, immune cell score, combined positive score), with higher expression linked to better response. Genetic biomarkers (tumor mutational burden, copy number variations and oncogenic drivers e.g., EGFR, KRAS, STK11 mutations) influence immune recognition. Epigenetic modifications (DNA methylation and histone modifications particularly via histone deacetylases like HDAC6 and HDAC8) present promising tools for predicting immunotherapy response in lung cancer. Transcriptomic markers related to IFNγ signaling, DNA damage repair genes and T cell effector genes, further contribute to immunotherapy outcomes.
Figure 1. Molecular biomarkers of immunotherapy response in lung cancer. PD-1 blockade with immune checkpoint inhibitors (ICIs) restores T cell activity against tumor cells by disrupting the interaction between PD-1 and PD-L1 on T and tumor cells, respectively. Key molecular biomarkers include PD-L1 expression, assessed through various scoring methods (tumor proportion score, tumor cell score, immune cell score, combined positive score), with higher expression linked to better response. Genetic biomarkers (tumor mutational burden, copy number variations and oncogenic drivers e.g., EGFR, KRAS, STK11 mutations) influence immune recognition. Epigenetic modifications (DNA methylation and histone modifications particularly via histone deacetylases like HDAC6 and HDAC8) present promising tools for predicting immunotherapy response in lung cancer. Transcriptomic markers related to IFNγ signaling, DNA damage repair genes and T cell effector genes, further contribute to immunotherapy outcomes.
CD8⁺ cytotoxic T cells are the core effector population responsible for antitumor immunity (Figure 2). One of the central effects of immune checkpoint inhibitors is to reinvigorate exhausted or functionally impaired CD8⁺ T cells [45]. This suggests that clinical benefit may depend not only on how many CD8⁺ T cells are present in the tumor microenvironment, but also on their activation status and functional capacity. In this context, assessment of CD8+ T-cell infiltration and activity is a reasonable strategy with suitable predictive relevance [46]. Similarly, scRNA-seq studies performed on patients undergoing combined immune checkpoint inhibitor and chemotherapy suggested that patients exhibiting a major pathological response showed more expansion of effector memory T cells, tissue-resident memory T cells and circulating effector T cells than nonresponders [47]. At the level of clinical outcome, a meta-analysis examining CD8+ tumor-infiltrating lymphocytes in patients treated with immune checkpoint inhibitors including samples of patients with NSCLC found an increase in CD8+ T-cell infiltration was associated with improved overall and progression-free survival, and objective response rates [48]. Consistent with these findings, a retrospective analysis found that in patients treated with chemoradiotherapy with the addition of immune checkpoint inhibitors, high baseline density of CD8+ tumor-infiltrating lymphocytes was significantly correlated with prolonged progression-free survival, but there was no prognostic impact in patients treated with chemoradiotherapy alone [49]. In anti-PD-1 treated metastatic NSCLC, objective response rates rose markedly as intratumoral CD8⁺ tumor-infiltrating lymphocyte density increased [50].
At the same time, wide differences in cutoff definitions across studies have made it difficult to standardize CD8⁺ T cells as predictive biomarkers in routine practice. A further complication is that CD8⁺ tumor-infiltrating lymphocytes are not uniformly functional: some populations may be poorly cytotoxic, non-tumor-reactive, or otherwise unresponsive to immune checkpoint inhibitor-mediated reinvigoration, and thus may contribute little to effective antitumor immunity [51, 52]. For this reason, relying only on total CD8⁺ T-cell numbers has not consistently predicted immune checkpoint inhibitor benefit in all cohorts. To overcome this limitation, recent work has increasingly focused on phenotypically defined CD8⁺ T-cell subsets. CD39, in particular, has been proposed as a marker enriched on tumor-specific T cells, and CD39⁺ CD8⁺ T cells have been reported as independent predictors of response to PD-1 or PD-L1 blockade in NSCLC, with responders showing significantly higher proportions of these cells than nonresponders [47]. Similarly, high PD-1 expression on CD8⁺ T cells reflects strong tumor antigen recognition and is closely linked to effective antitumor immunity. Responders to immune checkpoint inhibitors display significantly elevated PD-1 expression on CD8⁺ T cells relative to nonresponders [53]. Even though PD-1 expression is commonly interpreted as a feature of T-cell dysfunction or exhaustion, PD-1⁺ CD8⁺ tumor-infiltrating lymphocytes are still capable of sustained CXCL13 secretion. This chemokine can attract CXCR5-expressing B cells and follicular helper T cells, thereby supporting the development of tertiary lymphoid structures. In NSCLC, patients who respond to PD-1 blockade have a significantly higher fraction of PD-1 T cells, and this population is associated with better overall survival [54].
B cells
Tumor-infiltrating B cells drive local antitumor antibody production and the expansion of CD4⁺ T-cell clones [55]. By secreting cytokines, these B cells can further amplify effector T-cell function. Tumor-infiltrating B cells correlate with increased proportions of activated and memory CD4⁺ T cells and with lower Treg infiltration, supporting a role for these structures in organizing effective antitumor immunity and influencing prognosis (Figure 2) [56, 57]. Consistently, expression of B-cell-related gene programs, including CXCL13, has been linked to improved immunotherapy results in NSCLC patients receiving neoadjuvant immunochemotherapy [58]. Tumors from chemo + immunotherapy-treated patients achieving major pathological response show significantly greater B-cell infiltration than tumors from cases without major pathological response [59]. Further scRNA-seq work identified FCRL4⁺FCRL5⁺ B cells (memory), which are more abundant in patients with major pathological response. These cells may contribute to improved treatment efficacy and function as predictive biomarkers for immune checkpoint inhibitor responsiveness in NSCLC [60].
Immune-inhibitory cells
An immunosuppressive tumor microenvironment is sustained by multiple inhibitory cellular populations. This is further strengthened by suppressive cytokines including IL-6, IL-8 and TGF-b and is frequently linked to poorer responses to immunotherapy [61]. Among these factors, circulating immunosuppressive cells, particularly myeloid-derived suppressor cells, have been repeatedly linked with the outcome of immune checkpoint inhibitors, and may represent clinically useful predictive information [62]. At the same time, the intratumoral suppressive populations can directly remodel the immune activity in the tumor and as such can affect the efficacy of treatment. scRNA-seq analyses of samples from patients with lung cancer obtained before and during PD-1 blockade therapy have shown unique Treg dynamics in responders and nonresponders. Patients who do respond usually exhibit only a relatively small decrease in Treg frequency, while nonresponders exhibit a more significant increase in Treg frequency together with upregulation of immunosuppressive genes such as IL1R2, REL and LAYN [46]. Mechanistically, this observation is biologically plausible because immune checkpoint inhibitor therapy upregulates T-cell activation through shared signaling pathways that go through the T-cell receptor and CD28. As a result, immune checkpoint inhibitors may increase activation not only in effector CD8+ T cells but may also stimulate inhibitory Tregs. Under these conditions, therapeutic benefit is associated with the balance of these populations. Consistent with this concept, responders are characterized by high PD-1 expression predominantly on CD8⁺ T cells, while in nonresponders PD-1 expression is enriched on Tregs. Therefore, the ratio of PD-1high CD8⁺ T cells to PD-1high Tregs has been suggested as a useful predictive metric, with the most favorable responses observed when PD-1 expression is elevated in CD8⁺ T cells but relatively low in Tregs (Figure 2) [53]. Macrophage heterogeneity within the tumor microenvironment further influences immune checkpoint inhibitor responses. scRNA-seq profiling has identified Macro_SPP1 macrophages, which promote angiogenesis, and Macro_SELENOP macrophages, which exhibit anti-inflammatory properties. Both subsets display M2-like, tumor-promoting features characteristic of tumor-associated macrophages (Figure 2). Following immune checkpoint inhibitors therapy, Macro_SPP1 macrophages decrease, whereas Macro_SELENOP macrophages increase in patients without major pathological response. Moreover, cancer-associated fibroblast-associated gene signatures are significantly enriched in patients with progressive disease compared with those achieving complete or partial responses after anti-PD-1 therapy [63].
Tertiary lymphoid structures
During chronic inflammatory states, tertiary lymphoid structures can form ectopically in nonlymphoid tissues and develop both in terms of their architectural organization and their immunologic functions [64]. Their formation is orchestrated by chemokines and cytokines, which promote the recruitment of lymphocytes and myeloid cells [65]. Within tumors, checkpoint-like tertiary lymphoid structures can be present in stroma (extratumoral), tumor parenchyma (intratumoral), or at the invasive margin (peritumoral), mainly maturing in a continuum from rather loose collections of lymphocytes to lymphoid-like structures. Intratumoral tertiary lymphoid structure formation is biologically significant because it reduces dependence on immune-cell migration to distant secondary lymphoid organs. By functioning as a local immune “hub” near malignant tissue, Tertiary lymphoid structures may enable more rapid priming and expansion of T- and B-cell responses at the tumor site, potentially accelerating and strengthening antitumor immunity [66]. In cancer types, intratumoral Tertiary lymphoid structures are generally associated with improved treatment responses and reduced risk of cancer recurrence and thus support their relevance as both prognostic and predictive factors in immuno-oncology [65, 67]. These observations have also provided motivation for therapeutic strategies that aim to induce or boost tertiary lymphoid structure formation, either alone or together with immune checkpoint inhibitors, especially considering that immune checkpoint inhibitors themselves may induce tertiary lymphoid structure formation in certain contexts [68].
In lung adenocarcinoma, mature tertiary lymphoid structures are strongly associated with prolonged overall and disease-free survival, and their presence protects against metastasis (Figure 2) [69]. When NSCLC patients are further stratified by tertiary lymphoid structure maturity (low vs. high maturity), treatment-associated differences become more apparent. In one study, high-maturity tertiary lymphoid structures were identified in 30 of 40 patients treated with neoadjuvant immunochemotherapy, compared with 13 of 41 patients receiving chemotherapy alone and 25 of 40 untreated controls. This tendency helps to believe that chemotherapy can impair the maturation of tertiary lymphoid structure, but immune checkpoint inhibitors can stimulate the development of tertiary lymphoid structures [70]. The same analysis also showed a difference in clinical outcomes of treatment groups. Major pathological response occurred in 45.0% of patients who received neoadjuvant immunochemotherapy, including 14 cases of pathological complete response, whereas the chemotherapy-only group showed a major pathological response rate of 17.1% with only 2 cases achieving pathological complete response. Notably, tumors from patients achieving major pathological response had significantly higher tertiary lymphoid structure maturity, and tertiary lymphoid structure maturity served as an independent predictor of disease-free survival within the immunochemotherapy cohort [70]. Similar trends have been reported elsewhere, including longer disease-free survival in patients with high-maturity tertiary lymphoid structures compared with low-maturity Tertiary lymphoid structures [71]. After neoadjuvant immunotherapy, mature tertiary lymphoid structures have also been linked to higher CD8⁺ T-cell density and greater epithelial infiltration, consistent with enhanced immune activation in the tumor microenvironment [72]. Taken together, these data support a model in which neoadjuvant immunochemotherapy promotes tertiary lymphoid structure maturation, which is then associated with stronger antitumor immune responses and improved clinical outcomes.
Tertiary lymphoid structures density varies considerably among patients, and their formation has been reported more often in responders than in nonresponders after neoadjuvant immunochemotherapy, and tertiary lymphoid structure-positive cases showed significantly longer disease-free survival [73]. In addition, increases in tertiary lymphoid structure number and size after neoadjuvant immunotherapy were associated with improved long-term event-free survival [72]. Beyond simple presence/absence, higher tertiary lymphoid structure density has been associated with increased rates of pathological complete response and major pathological response in NSCLC patients treated with neoadjuvant immunochemotherapy [74]. Increased tertiary lymphoid structure density has also been observed in patients who achieved major pathological response and in responsive lesions from individuals with multiple primary lung cancers treated with neoadjuvant pembrolizumab [75]. Consistent with these observations, phase II clinical trial results further indicated that tertiary lymphoid structure abundance was significantly higher in patients with pathological complete response or major pathological response than in nonresponders [76]. Transcriptomic analyses after neoadjuvant durvalumab or pembrolizumab-based therapy revealed upregulation of tertiary lymphoid structure-related genes, including CXCL13, alongside increased tertiary lymphoid structure density by multiplex immunohistochemistry [77]. Collectively, these findings position Tertiary lymphoid structures as strong candidate biomarkers associated with pathological response and longer-term benefit from immune checkpoint inhibitors in NSCLC.
Tertiary lymphoid structures also show clear spatial heterogeneity in NSCLC, appearing intratumorally, peritumorally, or in extratumoral stromal regions [66]. Such spatial heterogeneity is presumably a biology of the underlying tumor and potentially can be of significance to outcomes. The intratumoral Tertiary lymphoid structures in NSCLC have been in many cases reported to be at higher density and frequency as compared to those at almost the invasive margin or in stroma surrounding the tumor. Significantly, tertiary lymphoid structure location has been linked to tumor aggressiveness, metastatic behavior and other clinicopathologic features. In some contexts, high densities of peritumoral or extratumoral tertiary lymphoid structures have been linked to invasive characteristics and worse prognosis. Similar site-dependent effects have also been described in other malignancies [78]. Together, these observations suggest that tertiary lymphoid structure localization may matter for predicting immunotherapy benefit and could eventually inform treatment planning. Conceptually, strategies that promote or leverage intratumoral tertiary lymphoid structures may be more effective at strengthening local immune priming and activation within the tumor bed, thereby improving therapeutic responses. However, given the limited number of studies addressing tertiary lymphoid structure spatial distribution in NSCLC, further systematic investigations are needed to clarify their precise roles.
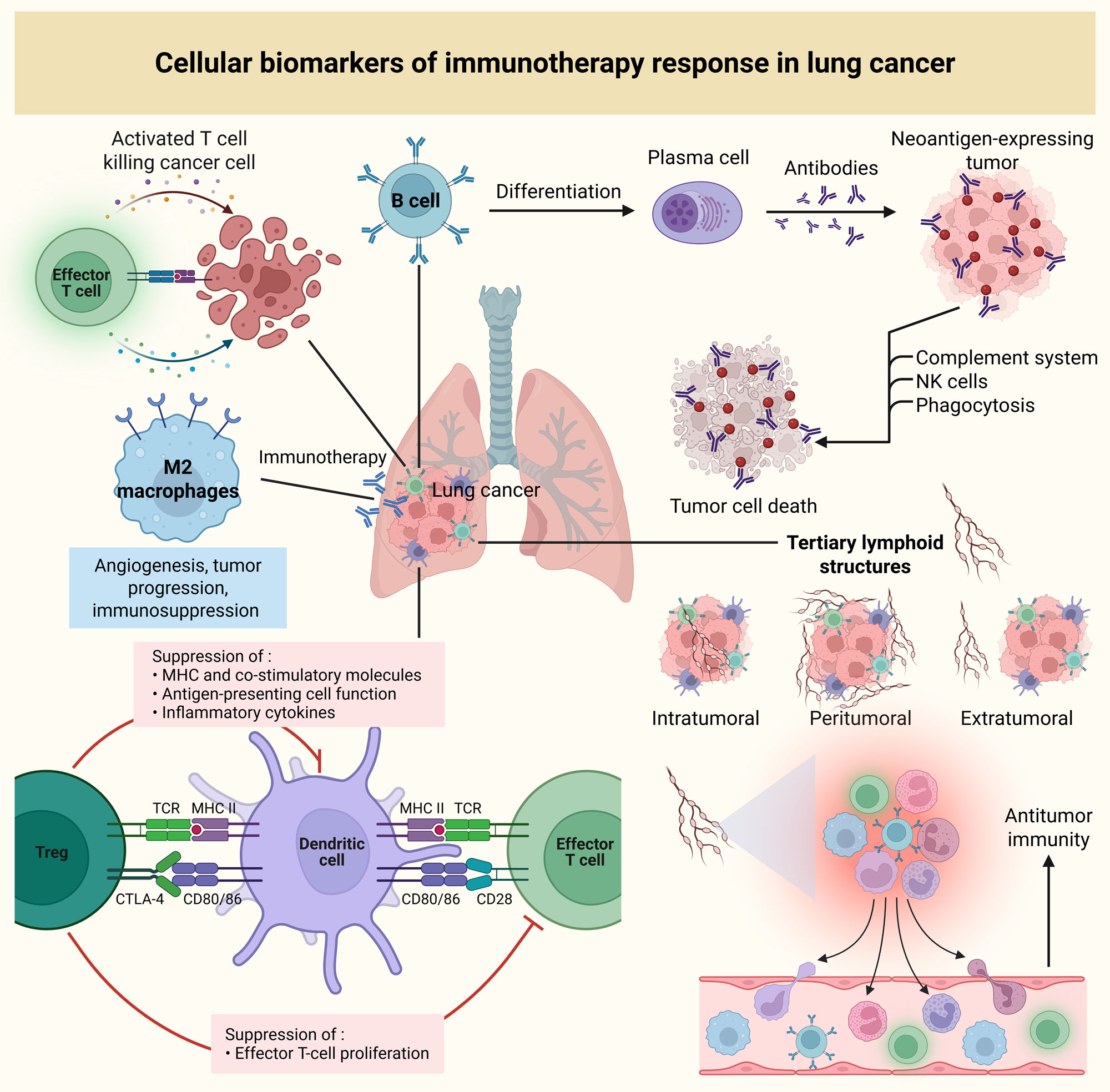 Figure 2. Cellular biomarkers of immunotherapy response in lung cancer. Activated CD8⁺ cytotoxic T cells, which are central to antitumor immunity, are rejuvenated by immune checkpoint inhibitors, driving enhanced tumor cell killing. Tumor-infiltrating B cells, through antibody production and cytokine secretion, contribute to T-cell activation and support antitumor responses. M2 macrophages, associated with immunosuppressive functions, promote tumor progression but may also alter following immunotherapy. Treg cells, which suppress effector T-cell activation, can also limit the efficacy of immunotherapy by downregulating immune responses. Tertiary lymphoid structures, which can develop intratumorally, peritumorally or extratumorally, are key indicators of immune response. The maturity and density of tertiary lymphoid structures, especially intratumoral structures, correlate with improved therapeutic responses and better survival outcomes.
Figure 2. Cellular biomarkers of immunotherapy response in lung cancer. Activated CD8⁺ cytotoxic T cells, which are central to antitumor immunity, are rejuvenated by immune checkpoint inhibitors, driving enhanced tumor cell killing. Tumor-infiltrating B cells, through antibody production and cytokine secretion, contribute to T-cell activation and support antitumor responses. M2 macrophages, associated with immunosuppressive functions, promote tumor progression but may also alter following immunotherapy. Treg cells, which suppress effector T-cell activation, can also limit the efficacy of immunotherapy by downregulating immune responses. Tertiary lymphoid structures, which can develop intratumorally, peritumorally or extratumorally, are key indicators of immune response. The maturity and density of tertiary lymphoid structures, especially intratumoral structures, correlate with improved therapeutic responses and better survival outcomes.
Circulating biomarkers are measurable tumor- or immune-related molecules present in blood or other body fluids, including ctDNA and extracellular vesicles, and offer a minimally invasive approach for diagnosis, prognostication, and therapeutic monitoring. Their clinical utility represents a major advance in precision oncology [79]. ctDNA refers to short DNA fragments released into the circulation from tumor cells and can capture tumor-specific genomic alterations. Both quantitative and qualitative features of ctDNA, such as mutation burden and epigenetic changes, may reflect immune checkpoint inhibitor response in NSCLC. Blood-based tumor mutational burden, estimated from ctDNA sequencing, has been developed as a proxy for tissue tumor mutational burden. In the MYSTIC trial, NSCLC patients with blood-based tumor mutational burden ≥20 mutations/Mb who received combination immunotherapy showed improved overall survival (Figure 3) [80]. Beyond mutational burden, absolute ctDNA abundance also appears informative. In the B-F1RST study of atezolizumab in NSCLC, patients with low ctDNA levels, defined by a maximum somatic allele fraction <1%, had substantially higher objective response rates than those with maximum somatic allele fraction ≥1% even though ctDNA quantity was insufficient for robust blood-based tumor mutational burden estimation in some cases [81]. In metastatic NSCLC, a ctDNA response defined as a >50% decrease in mutant allele fraction was associated with improved progression-free and overall survival and enabled earlier identification of patients likely to benefit from therapy [82]. Among patients with molecular residual disease after chemoradiotherapy who subsequently received consolidation immune checkpoint inhibitors, ctDNA trajectories separated clinical courses: rising ctDNA predicted rapid progression, whereas falling ctDNA levels were associated with benefit (Figure 3) [83]. Similarly, in NSCLC treated with immunochemotherapy, reductions in ctDNA allele fraction tracked with radiographic responses and favorable long-term outcomes, including higher response rates and improved progression-free and overall survival [84]. Altogether, these data support the prospects of ctDNA as an active biomarker of immune checkpoint inhibitor efficacy prediction and monitoring in NSCLC. Nevertheless, an insufficient number of standardized guidelines regarding the method of ctDNA collection, processing, and analysis along with the lack of large clinical validation of this approach to date limits its routine use in the treatment decision-making [85].
CTCs
CTCs, which disseminate from primary tumors into the bloodstream and may seed metastases, represent another potential biomarker source. The presence of CTCs, especially PD-L1-positive CTCs, in patients with advanced NSCLC who are treated with nivolumab was also linked to increased risk of disease progression (Figure 3), which still has to be confirmed by additional studies, as the populations in such studies are small [86]. Extracellular vesicles (EVs), including exosomes and microvesicles, are emerging as important mediators of intercellular communication and carriers of bioactive molecules [87]. Tumor-derived EVs have been explored as noninvasive biomarkers; higher expression of costimulatory or EV-associated markers such as CD9, CD81, and CD63 has been correlated with better objective response rate and immune checkpoint inhibitor responses [88]. EV-associated microRNAs may add predictive information as well; for example, EV-miR-625-5p has been reported as an independent predictor of immune checkpoint inhibitor response in NSCLC patients with PD-L1 expression ≥50% [89]. EVs also offer an alternative platform for PD-L1 assessment, since higher levels of exosomal PD-L1 have been associated with improved objective response rate and overall survival (Figure 3) [90]. In longitudinal analyses of melanoma and NSCLC treated with anti-PD-1 antibodies, plasma exosomal PD-L1 mRNA decreased significantly in responders at two months, remained relatively stable in patients with stable disease, and increased in those with progressive disease [91]. Emerging biosensor approaches, such as the Au SERP platform for quantitative detection of EV PD-1/PD-L1 mRNA, have shown encouraging performance in distinguishing responders from nonresponders, highlighting the translational potential of EV-based readouts [92].
Circulating immune cells
Populations of circulating immune cells are also predictive of responses to immune checkpoint inhibitors (Figure 3). Dynamic variations in the Tregs have been observed in patients with NSCLC receiving immune checkpoint blockade treatment and they were observed to be associated with specific clinical course [93]. The patients with pseudoprogression exhibited a significant decrease in circulating CD4⁺CD25⁺CD127lowFoxP3⁺ Tregs in a week after therapy initiation compared with hyperprogressive disease, which was linked to a significant rise. In responders, both total CD4⁺CD25⁺CD127lowFoxP3⁺ Tregs and PD-1-expressing CD4⁺CD25⁺CD127lowFoxP3⁺ Tregs declined significantly [94]. In another NSCLC cohort receiving anti-PD-1 antibodies, higher baseline frequencies of circulating CD4⁺CCR9⁺, CD4⁺CCR10⁺, or CD8⁺CXCR4⁺ T cells were associated with worse overall survival compared with lower baseline levels [95]. In advanced NSCLC treated with atezolizumab, patients with disease control showed an increased lymphocyte ratio, whereas those with progression demonstrated a significant decline. Progression was accompanied by reduced circulating CD4⁺ and CD8⁺ T cells and higher Tregs and myeloid-derived suppressor cells, while the inverse immune profile was observed in patients with disease control [96]. Among NSCLC patients treated with nivolumab, a higher ratio of central memory to effector T cells was associated with improved progression-free survival and correlated with higher tumor PD-L1 expression. In contrast, enrichment of exhausted T cells and depletion of memory effector CD8⁺ T cells were linked to disease progression [97].
Systemic inflammation indices
Systemic inflammatory indices derived from peripheral blood counts further reflect host immune status. The neutrophil-to-lymphocyte ratio, calculated from absolute circulating neutrophil and lymphocyte counts, is closely associated with innate immune activity and tumor burden (Figure 3) [98]. In NSCLC patients receiving neoadjuvant immune checkpoint inhibitors, a >10% reduction in neutrophil-to-lymphocyte ratio within four weeks was associated with tumor regression, major pathological response, and improved progression-free and overall survival [99, 100]. Similarly, in patients with PD-L1 TPS ≥50% and without EGFR or ALK alterations treated with pembrolizumab, a derived neutrophil-to-lymphocyte ratio <2.6 was linked to higher objective response rate and longer progression-free and overall survival compared with derived neutrophil-to-lymphocyte ratio ≥2.6 [101]. In advanced NSCLC treated with nivolumab, early increases in circulating cell-free DNA and neutrophil-to-lymphocyte ratio during the first six weeks were associated with poorer survival outcomes, suggesting a role for real-time monitoring of emerging resistance [102]. Additional ratios have also been evaluated: a higher monocyte-to-lymphocyte ratio and elevated absolute neutrophil counts have been associated with shorter progression-free and overall survival (Figure 3) [103], whereas lower platelet-to-lymphocyte ratios and monocyte-to-lymphocyte ratios are associated with improved progression-free survival [104].
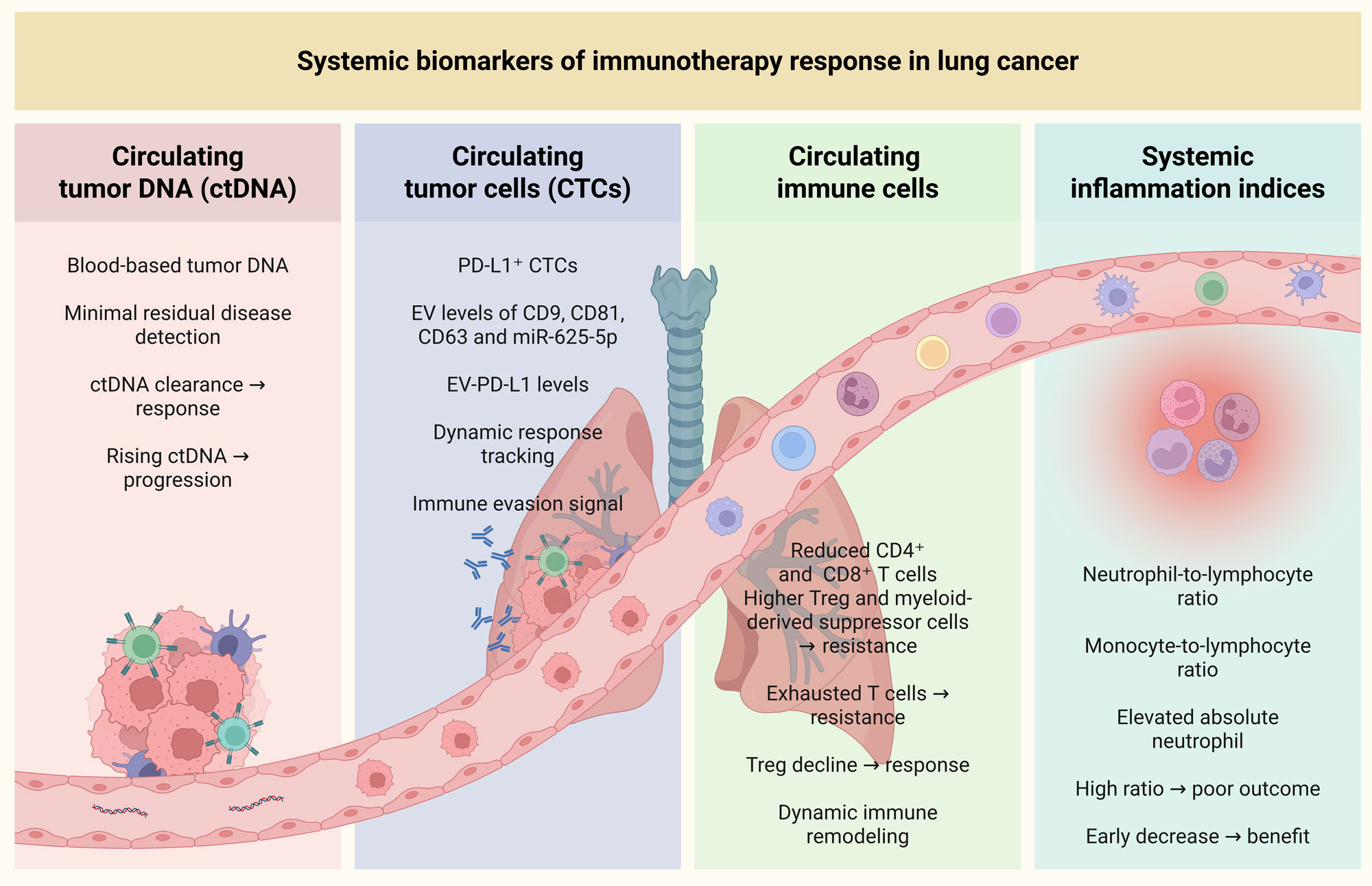 Figure 3. Cellular biomarkers of immunotherapy response in lung cancer. Circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), circulating immune cells, and systemic inflammation indices serve as biomarkers to monitor immune checkpoint inhibitor (ICI) responses in lung cancer. ctDNA, representing blood-based tumor DNA, offers insights into minimal residual disease, with rising ctDNA levels indicating disease progression and ctDNA clearance correlating with therapeutic response. CTCs, particularly PD-L1-positive CTCs, and extracellular vesicles (EV) are linked to immune evasion and can track dynamic responses to immunotherapy, with higher levels associated with poorer outcomes. Circulating immune cell subsets, such as reduced CD4⁺ and CD8⁺ T cells, and increased Tregs, myeloid-derived suppressor cells and exhausted T cells, are indicative of resistance to ICIs. In contrast, a decline in Tregs correlates with better responses. Systemic inflammation indices, including the neutrophil-to-lymphocyte ratio, monocyte-to-lymphocyte ratio and elevated absolute neutrophils provide additional prognostic information, with high ratios linked to poor outcomes and early decreases suggesting therapeutic benefits.
Figure 3. Cellular biomarkers of immunotherapy response in lung cancer. Circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), circulating immune cells, and systemic inflammation indices serve as biomarkers to monitor immune checkpoint inhibitor (ICI) responses in lung cancer. ctDNA, representing blood-based tumor DNA, offers insights into minimal residual disease, with rising ctDNA levels indicating disease progression and ctDNA clearance correlating with therapeutic response. CTCs, particularly PD-L1-positive CTCs, and extracellular vesicles (EV) are linked to immune evasion and can track dynamic responses to immunotherapy, with higher levels associated with poorer outcomes. Circulating immune cell subsets, such as reduced CD4⁺ and CD8⁺ T cells, and increased Tregs, myeloid-derived suppressor cells and exhausted T cells, are indicative of resistance to ICIs. In contrast, a decline in Tregs correlates with better responses. Systemic inflammation indices, including the neutrophil-to-lymphocyte ratio, monocyte-to-lymphocyte ratio and elevated absolute neutrophils provide additional prognostic information, with high ratios linked to poor outcomes and early decreases suggesting therapeutic benefits.
Tumor-infiltrating lymphocytes, especially CD8⁺ T cells, are central to effective antitumor immunity, and higher CD8⁺ T-cell infiltration is generally associated with improved responses to immunotherapy. In contrast, immunosuppressive compartments, including Tregs, tumor-associated neutrophils, tumor-associated macrophages, and cancer-associated fibroblasts, can enable immune escape and limit treatment benefits. Cancer-associated fibroblasts have traditionally been viewed as tumor-promoting because they support angiogenesis, invasion, and therapy resistance [111]. However, more recent single-cell and spatial transcriptomic work suggests that cancer-associated fibroblast biology is more complex, with certain cancer-associated fibroblast subsets potentially facilitating tertiary lymphoid structure formation, enhancing immune-cell recruitment, and supporting antitumor immune function [112, 113]. Response biology is also driven by genomic factors (especially by tumor mutational burden) that have a higher chance of generating neoantigens and being identified by T-cells. Nevertheless, even though tumor mutational burden is relatively high in NSCLC, even a minor part of nonsynonymous mutations translates into any neoantigen that can be considered immunogenic. Besides, tumor mutational burden does not have standardized thresholds, which weakens the value of tumor mutational burden as an independent clinical biomarker [114]. To this end, tumor mutational burden should be used in combination with other types of markers, including PD-L1 expression and systemic inflammatory indices, which may be more predictive [115]. In parallel, circulating biomarkers, including ctDNA kinetics, immune-cell subsets, and soluble proteins, provide minimally invasive options for prediction and longitudinal monitoring. Reduced ctDNA levels and consistent improvements in post-treatment inflammatory indices tend to follow clinical improvement across a wide range of studies, supplementary prognostic data may be found with soluble PD-L1, serum granzyme B, and so on, but findings remain inconsistent among cohorts [116].
Altogether, tumor-infiltrating lymphocytes and tertiary lymphoid structures offer an understanding of immune competence in the tumor microenvironment, a significant tumor immune evasion axis is mediated by PD-L1. Nevertheless, both types of biomarkers provide incomplete insights, and none of them can explain the complexity of immunotherapy response. Other sources of limitations are the lack of standardization of detection measures, and high inter-study heterogeneity. In the future, it is advisable to focus on the intense validation of predictive markers in very different patient groups, as well as the creation of multiparametric approaches. Integrative models that combine multiple biomarker classes, potentially enhanced through data-driven computational approaches, may better capture tumor-host interactions and improve response prediction. Advancing these strategies will be essential for reliably distinguishing responders from nonresponders and represents an important direction for the next phase of personalized immunotherapy in lung cancer.
No applicable.
Ethics approval
No applicable.
Data availability
The data will be available upon request.
Funding
None.
Authors’ contribution
Omnia Mohamed Attia performed the literature search, data collection, critical analysis of the included studies and masnuctipt writting.
Competing interests
The authors declare no competing interests.
- Herbst RS, Morgensztern D, Boshoff C: The biology and management of non-small cell lung cancer. Nature 2018, 553(7689): 446-454.
- Wang Y, Wang M, Wu HX, Xu RH: Advancing to the era of cancer immunotherapy. Cancer Commun (Lond) 2021, 41(9): 803-829.
- Lahiri A, Maji A, Potdar PD, Singh N, Parikh P, Bisht B, Mukherjee A, Paul MK: Lung cancer immunotherapy: progress, pitfalls, and promises. Mol Cancer 2023, 22(1): 40.
- Tufail M, Jiang CH, Li N: Immune evasion in cancer: mechanisms and cutting-edge therapeutic approaches. Signal Transduct Target Ther 2025, 10(1): 227.
- Racacho KJ, Shiau YP, Villa R, Mahri S, Tang M, Lin TY, Li Y: The tumor immune microenvironment: implications for cancer immunotherapy, treatment strategies, and monitoring approaches. Front Immunol 2025, 16: 1621812.
- Bai R, Lv Z, Xu D, Cui J: Predictive biomarkers for cancer immunotherapy with immune checkpoint inhibitors. Biomark Res 2020, 8: 34.
- Wang C, Wang HN, Wang L: Biomarkers for predicting the efficacy of immune checkpoint inhibitors. J Cancer 2022, 13(2): 481-495.
- Mariniello A, Borgeaud M, Weiner M, Frisone D, Kim F, Addeo A: Primary and Acquired Resistance to Immunotherapy with Checkpoint Inhibitors in NSCLC: From Bedside to Bench and Back. BioDrugs 2025, 39(2): 215-235.
- Fugel HJ, Nuijten M, Postma M: Stratified medicine and reimbursement issues. Front Pharmacol 2012, 3: 181.
- Bindal P, Gray JE, Boyle TA, Florou V, Puri S: Biomarkers of therapeutic response with immune checkpoint inhibitors. Ann Transl Med 2021, 9(12): 1040.
- Hegoburu A, Amer M, Frizelle F, Purcell R: B cells and tertiary lymphoid structures in cancer therapy response. BJC Rep 2025, 3(1): 40.
- Goh KY, Cheng TY, Tham SC, Lim DW: Circulating Biomarkers for Prediction of Immunotherapy Response in NSCLC. Biomedicines 2023, 11(2): 508.
- Lin X, Kang K, Chen P, Zeng Z, Li G, Xiong W, Yi M, Xiang B: Regulatory mechanisms of PD-1/PD-L1 in cancers. Mol Cancer 2024, 23(1): 108.
- Gainor JF, Rizvi H, Jimenez Aguilar E, Skoulidis F, Yeap BY, Naidoo J, Khosrowjerdi S, Mooradian M, Lydon C, Illei P et al: Clinical activity of programmed cell death 1 (PD-1) blockade in never, light, and heavy smokers with non-small-cell lung cancer and PD-L1 expression ≥50. Ann Oncol 2020, 31(3): 404-411.
- Doroshow DB, Bhalla S, Beasley MB, Sholl LM, Kerr KM, Gnjatic S, Wistuba, II, Rimm DL, Tsao MS, Hirsch FR: PD-L1 as a biomarker of response to immune-checkpoint inhibitors. Nat Rev Clin Oncol 2021, 18(6): 345-362.
- Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN et al: Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515(7528): 563-567.
- Topalian SL, Hodi FS, Brahmer JR, Gettinger SN, Smith DC, McDermott DF, Powderly JD, Carvajal RD, Sosman JA, Atkins MB et al: Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med 2012, 366(26): 2443-2454.
- Garon EB, Rizvi NA, Hui R, Leighl N, Balmanoukian AS, Eder JP, Patnaik A, Aggarwal C, Gubens M, Horn L et al: Pembrolizumab for the treatment of non-small-cell lung cancer. N Engl J Med 2015, 372(21): 2018-2028.
- Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ et al: Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016, 387(10027): 1540-1550.
- Reck M, Rodríguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S et al: Pembrolizumab versus Chemotherapy for PD-L1-Positive Non-Small-Cell Lung Cancer. N Engl J Med 2016, 375(19): 1823-1833.
- Pai-Scherf L, Blumenthal GM, Li H, Subramaniam S, Mishra-Kalyani PS, He K, Zhao H, Yu J, Paciga M, Goldberg KB et al: FDA Approval Summary: Pembrolizumab for Treatment of Metastatic Non-Small Cell Lung Cancer: First-Line Therapy and Beyond. Oncologist 2017, 22(11): 1392-1399.
- Brahmer JR, Lee JS, Ciuleanu TE, Bernabe Caro R, Nishio M, Urban L, Audigier-Valette C, Lupinacci L, Sangha R, Pluzanski A et al: Five-Year Survival Outcomes With Nivolumab Plus Ipilimumab Versus Chemotherapy as First-Line Treatment for Metastatic Non-Small-Cell Lung Cancer in CheckMate 227. J Clin Oncol 2023, 41(6): 1200-1212.
- Paz-Ares L, Spira A, Raben D, Planchard D, Cho BC, Özgüroğlu M, Daniel D, Villegas A, Vicente D, Hui R et al: Outcomes with durvalumab by tumour PD-L1 expression in unresectable, stage III non-small-cell lung cancer in the PACIFIC trial. Ann Oncol 2020, 31(6): 798-806.
- Mino-Kenudson M, Schalper K, Cooper W, Dacic S, Hirsch FR, Jain D, Lopez-Rios F, Tsao MS, Yatabe Y, Beasley MB et al: Predictive Biomarkers for Immunotherapy in Lung Cancer: Perspective From the International Association for the Study of Lung Cancer Pathology Committee. J Thorac Oncol 2022, 17(12): 1335-1354.
- Chan TA, Yarchoan M, Jaffee E, Swanton C, Quezada SA, Stenzinger A, Peters S: Development of tumor mutation burden as an immunotherapy biomarker: utility for the oncology clinic. Ann Oncol 2019, 30(1): 44-56.
- Rizvi NA, Hellmann MD, Snyder A, Kvistborg P, Makarov V, Havel JJ, Lee W, Yuan J, Wong P, Ho TS et al: Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 2015, 348(6230): 124-128.
- Carbone DP, Reck M, Paz-Ares L, Creelan B, Horn L, Steins M, Felip E, van den Heuvel MM, Ciuleanu TE, Badin F et al: First-Line Nivolumab in Stage IV or Recurrent Non-Small-Cell Lung Cancer. N Engl J Med 2017, 376(25): 2415-2426.
- Goldberg SB, Narayan A, Kole AJ, Decker RH, Teysir J, Carriero NJ, Lee A, Nemati R, Nath SK, Mane SM et al: Early Assessment of Lung Cancer Immunotherapy Response via Circulating Tumor DNA. Clin Cancer Res 2018, 24(8): 1872-1880.
- Otano I, Ucero AC, Zugazagoitia J, Paz-Ares L: At the crossroads of immunotherapy for oncogene-addicted subsets of NSCLC. Nat Rev Clin Oncol 2023, 20(3): 143-159.
- Ramos-Ramírez M, Caballe-Pérez E, Lucio-Lozada J, Romero-Nuñez E, Castillo-Ruiz C, Dorantes-Sánchez L, Flores-Estrada D, Recondo G, Barrios-Bernal P, Cabrera-Miranda L et al: Immunomodulatory role of oncogenic alterations in non-small cell lung cancer: a review of implications for immunotherapy. Cancer Metastasis Rev 2025, 44(1): 30.
- Zhang H, Pang Y, Yi L, Wang X, Wei P, Wang H, Lin S: Epigenetic regulators combined with tumour immunotherapy: current status and perspectives. Clin Epigenetics 2025, 17(1): 51.
- Yao X, Gao C, Sun C, Chen ZS, Zhuang J: Epigenetic code underlying EGFR-TKI resistance in non-small cell lung cancer: Elucidation of mechanisms and perspectives on therapeutic strategies. Drug Discov Today 2025, 30(3): 104321.
- Kim JY, Choi JK, Jung H: Genome-wide methylation patterns predict clinical benefit of immunotherapy in lung cancer. Clin Epigenetics 2020, 12(1): 119.
- Duruisseaux M, Martínez-Cardús A, Calleja-Cervantes ME, Moran S, Castro de Moura M, Davalos V, Piñeyro D, Sanchez-Cespedes M, Girard N, Brevet M et al: Epigenetic prediction of response to anti-PD-1 treatment in non-small-cell lung cancer: a multicentre, retrospective analysis. Lancet Respir Med 2018, 6(10): 771-781.
- Liu S, Ma S, Liu G, Hou L, Guan Y, Liu L, Meng Y, Yu W, Liu T, Zhou L et al: CK2B Induces CD8(+) T-Cell Exhaustion through HDAC8-Mediated Epigenetic Reprogramming to Limit the Efficacy of Anti-PD-1 Therapy in Non-Small-Cell Lung Cancer. Adv Sci (Weinh) 2025, 12(16): e2411053.
- Shin HS, Choi J, Lee J, Lee SY: Histone Deacetylase as a Valuable Predictive Biomarker and Therapeutic Target in Immunotherapy for Non-Small Cell Lung Cancer. Cancer Res Treat 2022, 54(2): 458-468.
- Lianidou E: Detection and relevance of epigenetic markers on ctDNA: recent advances and future outlook. Mol Oncol 2021, 15(6): 1683-1700.
- Chandra Sekar PK, Veerabathiran R: Systems immunology meets clinical translation: Multi-omic approaches to predict therapy response in cancer and autoimmune disease. Clin Immunol Commun 2026, 9: 12-22.
- Ayers M, Lunceford J, Nebozhyn M, Murphy E, Loboda A, Kaufman DR, Albright A, Cheng JD, Kang SP, Shankaran V et al: IFN-γ-related mRNA profile predicts clinical response to PD-1 blockade. J Clin Invest 2017, 127(8): 2930-2940.
- Fehrenbacher L, Spira A, Ballinger M, Kowanetz M, Vansteenkiste J, Mazieres J, Park K, Smith D, Artal-Cortes A, Lewanski C et al: Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet 2016, 387(10030): 1837-1846.
- Karachaliou N, Gonzalez-Cao M, Crespo G, Drozdowskyj A, Aldeguer E, Gimenez-Capitan A, Teixido C, Molina-Vila MA, Viteri S, De Los Llanos Gil M et al: Interferon gamma, an important marker of response to immune checkpoint blockade in non-small cell lung cancer and melanoma patients. Ther Adv Med Oncol 2018, 10: 1758834017749748.
- Lei Y, Zhang G, Zhang C, Xue L, Yang Z, Lu Z, Huang J, Zang R, Che Y, Mao S et al: The average copy number variation (CNVA) of chromosome fragments is a potential surrogate for tumor mutational burden in predicting responses to immunotherapy in non-small-cell lung cancer. Clin Transl Immunology 2021, 10(1): e1231.
- Qi X, Qi C, Wu T, Hu Y: CSF1R and HCST: Novel Candidate Biomarkers Predicting the Response to Immunotherapy in Non-Small Cell Lung Cancer. Technol Cancer Res Treat 2020, 19: 1533033820970663.
- Wang Y, Liu Y, Li X, Li W, Xue Z, He X, Xiong W, He L, Bai Y: TCR Coexpression Signature Predicts Immunotherapy Resistance in NSCLC. Front Pharmacol 2022, 13: 875149.
- Kumar S, Singh SK, Rana B, Rana A: Tumor-infiltrating CD8(+) T cell antitumor efficacy and exhaustion: molecular insights. Drug Discov Today 2021, 26(4): 951-967.
- Liu B, Hu X, Feng K, Gao R, Xue Z, Zhang S, Zhang Y, Corse E, Hu Y, Han W et al: Temporal single-cell tracing reveals clonal revival and expansion of precursor exhausted T cells during anti-PD-1 therapy in lung cancer. Nat Cancer 2022, 3(1): 108-121.
- Yeong J, Suteja L, Simoni Y, Lau KW, Tan AC, Li HH, Lim S, Loh JH, Wee FYT, Nerurkar SN et al: Intratumoral CD39(+)CD8(+) T Cells Predict Response to Programmed Cell Death Protein-1 or Programmed Death Ligand-1 Blockade in Patients With NSCLC. J Thorac Oncol 2021, 16(8): 1349-1358.
- Li F, Li C, Cai X, Xie Z, Zhou L, Cheng B, Zhong R, Xiong S, Li J, Chen Z et al: The association between CD8+ tumor-infiltrating lymphocytes and the clinical outcome of cancer immunotherapy: A systematic review and meta-analysis. EClinicalMedicine 2021, 41: 101134.
- Shirasawa M, Yoshida T, Imabayashi T, Okuma K, Matsumoto Y, Masuda K, Shinno Y, Okuma Y, Goto Y, Horinouchi H et al: Baseline PD-L1 expression and tumour-infiltrated lymphocyte status predict the efficacy of durvalumab consolidation therapy after chemoradiotherapy in unresectable locally advanced patients with non-small-cell lung cancer. Eur J Cancer 2022, 162: 1-10.
- Uryvaev A, Passhak M, Hershkovits D, Sabo E, Bar-Sela G: The role of tumor-infiltrating lymphocytes (TILs) as a predictive biomarker of response to anti-PD1 therapy in patients with metastatic non-small cell lung cancer or metastatic melanoma. Med Oncol 2018, 35(3): 25.
- Horton BL, Morgan DM, Momin N, Zagorulya M, Torres-Mejia E, Bhandarkar V, Wittrup KD, Love JC, Spranger S: Lack of CD8(+) T cell effector differentiation during priming mediates checkpoint blockade resistance in non-small cell lung cancer. Sci Immunol 2021, 6(64): eabi8800.
- Gueguen P, Metoikidou C, Dupic T, Lawand M, Goudot C, Baulande S, Lameiras S, Lantz O, Girard N, Seguin-Givelet A et al: Contribution of resident and circulating precursors to tumor-infiltrating CD8(+) T cell populations in lung cancer. Sci Immunol 2021, 6(55): eabd5778.
- Kumagai S, Togashi Y, Kamada T, Sugiyama E, Nishinakamura H, Takeuchi Y, Vitaly K, Itahashi K, Maeda Y, Matsui S et al: The PD-1 expression balance between effector and regulatory T cells predicts the clinical efficacy of PD-1 blockade therapies. Nat Immunol 2020, 21(11): 1346-1358.
- Thommen DS, Koelzer VH, Herzig P, Roller A, Trefny M, Dimeloe S, Kiialainen A, Hanhart J, Schill C, Hess C et al: A transcriptionally and functionally distinct PD-1(+) CD8(+) T cell pool with predictive potential in non-small-cell lung cancer treated with PD-1 blockade. Nat Med 2018, 24(7): 994-1004.
- Wang SS, Liu W, Ly D, Xu H, Qu L, Zhang L: Tumor-infiltrating B cells: their role and application in anti-tumor immunity in lung cancer. Cell Mol Immunol 2019, 16(1): 6-18.
- Germain C, Devi-Marulkar P, Knockaert S, Biton J, Kaplon H, Letaïef L, Goc J, Seguin-Givelet A, Gossot D, Girard N et al: Tertiary Lymphoid Structure-B Cells Narrow Regulatory T Cells Impact in Lung Cancer Patients. Front Immunol 2021, 12: 626776.
- Pradhan M, Chocry M, Gibbons DL, Sepesi B, Cascone T: Emerging biomarkers for neoadjuvant immune checkpoint inhibitors in operable non-small cell lung cancer. Transl Lung Cancer Res 2021, 10(1): 590-606.
- Cascone T, Leung CH, Weissferdt A, Pataer A, Carter BW, Godoy MCB, Feldman H, William WN, Jr., Xi Y, Basu S et al: Neoadjuvant chemotherapy plus nivolumab with or without ipilimumab in operable non-small cell lung cancer: the phase 2 platform NEOSTAR trial. Nat Med 2023, 29(3): 593-604.
- Hui Z, Zhang J, Ren Y, Li X, Yan C, Yu W, Wang T, Xiao S, Chen Y, Zhang R et al: Single-cell profiling of immune cells after neoadjuvant pembrolizumab and chemotherapy in IIIA non-small cell lung cancer (NSCLC). Cell Death Dis 2022, 13(7): 607.
- Hu J, Zhang L, Xia H, Yan Y, Zhu X, Sun F, Sun L, Li S, Li D, Wang J et al: Tumor microenvironment remodeling after neoadjuvant immunotherapy in non-small cell lung cancer revealed by single-cell RNA sequencing. Genome Med 2023, 15(1): 14.
- Zou J, Wang S, Zhang Y, Tian W, Mai G, Xu Y, Xiao W, Graves EE, Wu F: Tumor-educated cells in tumor microenvironment: Key drivers of immunotherapy resistance. Chin J Cancer Res 2025, 37(3): 446-465.
- Koh J, Kim Y, Lee KY, Hur JY, Kim MS, Kim B, Cho HJ, Lee YC, Bae YH, Ku BM et al: MDSC subtypes and CD39 expression on CD8(+) T cells predict the efficacy of anti-PD-1 immunotherapy in patients with advanced NSCLC. Eur J Immunol 2020, 50(11): 1810-1819.
- Galbo PM, Jr., Zang X, Zheng D: Molecular Features of Cancer-associated Fibroblast Subtypes and their Implication on Cancer Pathogenesis, Prognosis, and Immunotherapy Resistance. Clin Cancer Res 2021, 27(9): 2636-2647.
- Sautès-Fridman C, Petitprez F, Calderaro J, Fridman WH: Tertiary lymphoid structures in the era of cancer immunotherapy. Nat Rev Cancer 2019, 19(6): 307-325.
- Munoz-Erazo L, Rhodes JL, Marion VC, Kemp RA: Tertiary lymphoid structures in cancer - considerations for patient prognosis. Cell Mol Immunol 2020, 17(6): 570-575.
- Schumacher TN, Thommen DS: Tertiary lymphoid structures in cancer. Science 2022, 375(6576): eabf9419.
- Gago da Graça C, van Baarsen LGM, Mebius RE: Tertiary Lymphoid Structures: Diversity in Their Development, Composition, and Role. J Immunol 2021, 206(2): 273-281.
- Sato Y, Silina K, van den Broek M, Hirahara K, Yanagita M: The roles of tertiary lymphoid structures in chronic diseases. Nat Rev Nephrol 2023, 19(8): 525-537.
- Wakasu S, Tagawa T, Haratake N, Kinoshita F, Oku Y, Ono Y, Takenaka T, Oda Y, Shimokawa M, Mori M: Preventive effect of tertiary lymphoid structures on lymph node metastasis of lung adenocarcinoma. Cancer Immunol Immunother 2023, 72(6): 1823-1834.
- Sun X, Liu W, Sun L, Mo H, Feng Y, Wu X, Li C, Chen C, Li J, Xin Y et al: Maturation and abundance of tertiary lymphoid structures are associated with the efficacy of neoadjuvant chemoimmunotherapy in resectable non-small cell lung cancer. J Immunother Cancer 2022, 10(11): e005531.
- Liu Y, Xiong L, Chen Y, Cai R, Xu X, Wang T, Guo Y, Ruan L, Fang Y, Zhang Q et al: Complete pathological remission and tertiary lymphoid structures are associated with the efficacy of resectable NSCLC receiving neoadjuvant chemoimmunotherapy: A double-center retrospective study. Hum Vaccin Immunother 2023, 19(3): 2285902.
- Rothschild SI, Zippelius A, Eboulet EI, Savic Prince S, Betticher D, Bettini A, Früh M, Joerger M, Lardinois D, Gelpke H et al: SAKK 16/14: Durvalumab in Addition to Neoadjuvant Chemotherapy in Patients With Stage IIIA(N2) Non-Small-Cell Lung Cancer-A Multicenter Single-Arm Phase II Trial. J Clin Oncol 2021, 39(26): 2872-2880.
- Xu F, Zhu H, Dong Y, Li L, Liu N, Yuan S: Combined inflammatory parameters and tertiary lymphoid structure predict prognosis in patients with resectable non-small cell lung cancer treated with neoadjuvant chemoimmunotherapy. Front Immunol 2023, 14: 1244256.
- Cai W, Jing M, Gu Y, Bei T, Zhao X, Chen S, Wen J, Gao J, Wu C, Xue Z: Tumor microenvironment features decipher the outperformance of neoadjuvant immunochemotherapy over chemotherapy in resectable non-small cell lung cancer. Front Immunol 2022, 13: 984666.
- Zhang C, Yin K, Liu SY, Yan LX, Su J, Wu YL, Zhang XC, Zhong WZ, Yang XN: Multiomics analysis reveals a distinct response mechanism in multiple primary lung adenocarcinoma after neoadjuvant immunotherapy. J Immunother Cancer 2021, 9(4): e002312.
- Cottrell TR, Thompson ED, Forde PM, Stein JE, Duffield AS, Anagnostou V, Rekhtman N, Anders RA, Cuda JD, Illei PB et al: Pathologic features of response to neoadjuvant anti-PD-1 in resected non-small-cell lung carcinoma: a proposal for quantitative immune-related pathologic response criteria (irPRC). Ann Oncol 2018, 29(8): 1853-1860.
- Cascone T, Kar G, Spicer JD, García-Campelo R, Weder W, Daniel DB, Spigel DR, Hussein M, Mazieres J, Oliveira J et al: Neoadjuvant Durvalumab Alone or Combined with Novel Immuno-Oncology Agents in Resectable Lung Cancer: The Phase II NeoCOAST Platform Trial. Cancer Discov 2023, 13(11): 2394-2411.
- Xin S, Wen S, He P, Zhao Y, Zhao H: Density of tertiary lymphoid structures and their correlation with prognosis in non-small cell lung cancer. Front Immunol 2024, 15: 1423775.
- You W, Shang B, Sun J, Liu X, Su L, Jiang S: Mechanistic insight of predictive biomarkers for antitumor PD‑1/PD‑L1 blockade: A paradigm shift towards immunome evaluation (Review). Oncol Rep 2020, 44(2): 424-437.
- Rizvi NA, Cho BC, Reinmuth N, Lee KH, Luft A, Ahn MJ, van den Heuvel MM, Cobo M, Vicente D, Smolin A et al: Durvalumab With or Without Tremelimumab vs Standard Chemotherapy in First-line Treatment of Metastatic Non-Small Cell Lung Cancer: The MYSTIC Phase 3 Randomized Clinical Trial. JAMA Oncol 2020, 6(5): 661-674.
- Kang DH, Chung C, Sun P, Lee DH, Lee SI, Park D, Koh JS, Kim Y, Yi HS, Lee JE: Circulating regulatory T cells predict efficacy and atypical responses in lung cancer patients treated with PD-1/PD-L1 inhibitors. Cancer Immunol Immunother 2022, 71(3): 579-588.
- Moding EJ, Liu Y, Nabet BY, Chabon JJ, Chaudhuri AA, Hui AB, Bonilla RF, Ko RB, Yoo CH, Gojenola L et al: Circulating Tumor DNA Dynamics Predict Benefit from Consolidation Immunotherapy in Locally Advanced Non-Small Cell Lung Cancer. Nat Cancer 2020, 1(2): 176-183.
- Ricciuti B, Jones G, Severgnini M, Alessi JV, Recondo G, Lawrence M, Forshew T, Lydon C, Nishino M, Cheng M et al: Early plasma circulating tumor DNA (ctDNA) changes predict response to first-line pembrolizumab-based therapy in non-small cell lung cancer (NSCLC). J Immunother Cancer 2021, 9(3): e001504.
- Snyder A, Morrissey MP, Hellmann MD: Use of Circulating Tumor DNA for Cancer Immunotherapy. Clin Cancer Res 2019, 25(23): 6909-6915.
- Khagi Y, Goodman AM, Daniels GA, Patel SP, Sacco AG, Randall JM, Bazhenova LA, Kurzrock R: Hypermutated Circulating Tumor DNA: Correlation with Response to Checkpoint Inhibitor-Based Immunotherapy. Clin Cancer Res 2017, 23(19): 5729-5736.
- Zheng D, Liwinski T, Elinav E: Interaction between microbiota and immunity in health and disease. Cell Res 2020, 30(6): 492-506.
- Becker A, Thakur BK, Weiss JM, Kim HS, Peinado H, Lyden D: Extracellular Vesicles in Cancer: Cell-to-Cell Mediators of Metastasis. Cancer Cell 2016, 30(6): 836-848.
- Signorelli D, Ghidotti P, Proto C, Brambilla M, De Toma A, Ferrara R, Galli G, Ganzinelli M, Lo Russo G, Prelaj A et al: Circulating CD81-expressing extracellular vesicles as biomarkers of response for immune-checkpoint inhibitors in advanced NSCLC. Front Immunol 2022, 13: 987639.
- Pantano F, Zalfa F, Iuliani M, Simonetti S, Manca P, Napolitano A, Tiberi S, Russano M, Citarella F, Foderaro S et al: Large-Scale Profiling of Extracellular Vesicles Identified miR-625-5p as a Novel Biomarker of Immunotherapy Response in Advanced Non-Small-Cell Lung Cancer Patients. Cancers (Basel) 2022, 14(10): 2435.
- Yang Q, Chen M, Gu J, Niu K, Zhao X, Zheng L, Xu Z, Yu Y, Li F, Meng L et al: Novel Biomarkers of Dynamic Blood PD-L1 Expression for Immune Checkpoint Inhibitors in Advanced Non-Small-Cell Lung Cancer Patients. Front Immunol 2021, 12: 665133.
- Okuma Y, Hosomi Y, Nakahara Y, Watanabe K, Sagawa Y, Homma S: High plasma levels of soluble programmed cell death ligand 1 are prognostic for reduced survival in advanced lung cancer. Lung Cancer 2017, 104: 1-6.
- Nguyen LTH, Zhang J, Rima XY, Wang X, Kwak KJ, Okimoto T, Amann J, Yoon MJ, Shukuya T, Chiang CL et al: An immunogold single extracellular vesicular RNA and protein ((Au) SERP) biochip to predict responses to immunotherapy in non-small cell lung cancer patients. J Extracell Vesicles 2022, 11(9): e12258.
- Marcos Rubio A, Everaert C, Van Damme E, De Preter K, Vermaelen K: Circulating immune cell dynamics as outcome predictors for immunotherapy in non-small cell lung cancer. J Immunother Cancer 2023, 11(8): e007023.
- Rogado J, Pozo F, Troule K, Sánchez-Torres JM, Romero-Laorden N, Mondejar R, Donnay O, Ballesteros A, Pacheco-Barcia V, Aspa J et al: Peripheral Blood Mononuclear Cells Predict Therapeutic Efficacy of Immunotherapy in NSCLC. Cancers (Basel) 2022, 14(12): 2898.
- Zhuo M, Chen H, Zhang T, Yang X, Zhong J, Wang Y, An T, Wu M, Wang Z, Huang J et al: The potential predictive value of circulating immune cell ratio and tumor marker in atezolizumab treated advanced non-small cell lung cancer patients. Cancer Biomark 2018, 22(3): 467-476.
- Mosca M, Nigro MC, Pagani R, De Giglio A, Di Federico A: Neutrophil-to-Lymphocyte Ratio (NLR) in NSCLC, Gastrointestinal, and Other Solid Tumors: Immunotherapy and Beyond. Biomolecules 2023, 13(12): 1803.
- Kim CG, Kim KH, Pyo KH, Xin CF, Hong MH, Ahn BC, Kim Y, Choi SJ, Yoon HI, Lee JG et al: Hyperprogressive disease during PD-1/PD-L1 blockade in patients with non-small-cell lung cancer. Ann Oncol 2019, 30(7): 1104-1113.
- Hwang M, Canzoniero JV, Rosner S, Zhang G, White JR, Belcaid Z, Cherry C, Balan A, Pereira G, Curry A et al: Peripheral blood immune cell dynamics reflect antitumor immune responses and predict clinical response to immunotherapy. J Immunother Cancer 2022, 10(6): e004688.
- Ayers KL, Ma M, Debussche G, Corrigan D, McCafferty J, Lee K, Newman S, Zhou X, Hirsch FR, Mack PC et al: A composite biomarker of neutrophil-lymphocyte ratio and hemoglobin level correlates with clinical response to PD-1 and PD-L1 inhibitors in advanced non-small cell lung cancers. BMC Cancer 2021, 21(1): 441.
- Sánchez-Gastaldo A, Muñoz-Fuentes MA, Molina-Pinelo S, Alonso-García M, Boyero L, Bernabé-Caro R: Correlation of peripheral blood biomarkers with clinical outcomes in NSCLC patients with high PD-L1 expression treated with pembrolizumab. Transl Lung Cancer Res 2021, 10(6): 2509-2522.
- Passiglia F, Galvano A, Castiglia M, Incorvaia L, Calò V, Listì A, Mazzarisi S, Perez A, Gallina G, Rizzo S et al: Monitoring blood biomarkers to predict nivolumab effectiveness in NSCLC patients. Ther Adv Med Oncol 2019, 11: 1758835919839928.
- Del Re M, Marconcini R, Pasquini G, Rofi E, Vivaldi C, Bloise F, Restante G, Arrigoni E, Caparello C, Bianco MG et al: PD-L1 mRNA expression in plasma-derived exosomes is associated with response to anti-PD-1 antibodies in melanoma and NSCLC. Br J Cancer 2018, 118(6): 820-824.
- Soyano AE, Dholaria B, Marin-Acevedo JA, Diehl N, Hodge D, Luo Y, Manochakian R, Chumsri S, Adjei A, Knutson KL et al: Peripheral blood biomarkers correlate with outcomes in advanced non-small cell lung Cancer patients treated with anti-PD-1 antibodies. J Immunother Cancer 2018, 6(1): 129.
- Jiang M, Peng W, Pu X, Chen B, Li J, Xu F, Liu L, Xu L, Xu Y, Cao J et al: Peripheral Blood Biomarkers Associated With Outcome in Non-small Cell Lung Cancer Patients Treated With Nivolumab and Durvalumab Monotherapy. Front Oncol 2020, 10: 913.
- Zou Y, Hu X, Zheng S, Yang A, Li X, Tang H, Kong Y, Xie X: Discordance of immunotherapy response predictive biomarkers between primary lesions and paired metastases in tumours: A systematic review and meta-analysis. EBioMedicine 2021, 63: 103137.
- Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, Antonia S, Pluzanski A, Vokes EE, Holgado E et al: Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N Engl J Med 2015, 373(2): 123-135.
- Dall'Olio FG, Calabrò D, Conci N, Argalia G, Marchese PV, Fabbri F, Fragomeno B, Ricci D, Fanti S, Ambrosini V et al: Baseline total metabolic tumour volume on 2-deoxy-2-[18F]fluoro-d-glucose positron emission tomography-computed tomography as a promising biomarker in patients with advanced non-small cell lung cancer treated with first-line pembrolizumab. Eur J Cancer 2021, 150: 99-107.
- Niemeijer AN, Leung D, Huisman MC, Bahce I, Hoekstra OS, van Dongen G, Boellaard R, Du S, Hayes W, Smith R et al: Whole body PD-1 and PD-L1 positron emission tomography in patients with non-small-cell lung cancer. Nat Commun 2018, 9(1): 4664.
- Guibert N, Delaunay M, Lusque A, Boubekeur N, Rouquette I, Clermont E, Mourlanette J, Gouin S, Dormoy I, Favre G et al: PD-L1 expression in circulating tumor cells of advanced non-small cell lung cancer patients treated with nivolumab. Lung Cancer 2018, 120: 108-112.
- Dall'Olio FG, Gelsomino F, Conci N, Marcolin L, De Giglio A, Grilli G, Sperandi F, Fontana F, Terracciano M, Fragomeno B et al: PD-L1 Expression in Circulating Tumor Cells as a Promising Prognostic Biomarker in Advanced Non-small-cell Lung Cancer Treated with Immune Checkpoint Inhibitors. Clin Lung Cancer 2021, 22(5): 423-431.
- Zhang H, Yue X, Chen Z, Liu C, Wu W, Zhang N, Liu Z, Yang L, Jiang Q, Cheng Q et al: Define cancer-associated fibroblasts (CAFs) in the tumor microenvironment: new opportunities in cancer immunotherapy and advances in clinical trials. Mol Cancer 2023, 22(1): 159.
- Zhang Y, Liu G, Zeng Q, Wu W, Lei K, Zhang C, Tang M, Zhang Y, Xiang X, Tan L et al: CCL19-producing fibroblasts promote tertiary lymphoid structure formation enhancing anti-tumor IgG response in colorectal cancer liver metastasis. Cancer Cell 2024, 42(8): 1370-1385.e1379.
- Onder L, Papadopoulou C, Lütge A, Cheng HW, Lütge M, Perez-Shibayama C, Gil-Cruz C, De Martin A, Kurz L, Cadosch N et al: Fibroblastic reticular cells generate protective intratumoral T cell environments in lung cancer. Cell 2025, 188(2): 430-446.e420.
- Yadav M, Jhunjhunwala S, Phung QT, Lupardus P, Tanguay J, Bumbaca S, Franci C, Cheung TK, Fritsche J, Weinschenk T et al: Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature 2014, 515(7528): 572-576.
- Kao C, Powers E, Wu Y, Datto MB, Green MF, Strickler JH, Ready NE, Zhang T, Clarke JM: Predictive Value of Combining Biomarkers for Clinical Outcomes in Advanced Non-Small Cell Lung Cancer Patients Receiving Immune Checkpoint Inhibitors. Clin Lung Cancer 2021, 22(6): 500-509.
- Ando K, Hamada K, Watanabe M, Ohkuma R, Shida M, Onoue R, Kubota Y, Matsui H, Ishiguro T, Hirasawa Y et al: Plasma Levels of Soluble PD-L1 Correlate With Tumor Regression in Patients With Lung and Gastric Cancer Treated With Immune Checkpoint Inhibitors. Anticancer Res 2019, 39(9): 5195-5201.
Asia-Pacific Journal of Oncology
print ISSN: 2708-7980, online ISSN: 2708-7999
 Copyright © Asia Pac J Oncol. This work is licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International (CC BY-NC-ND 4.0) License.
Copyright © Asia Pac J Oncol. This work is licensed under a Creative Commons Attribution-NonCommercial-No Derivatives 4.0 International (CC BY-NC-ND 4.0) License.

 Submit Manuscript
Submit Manuscript